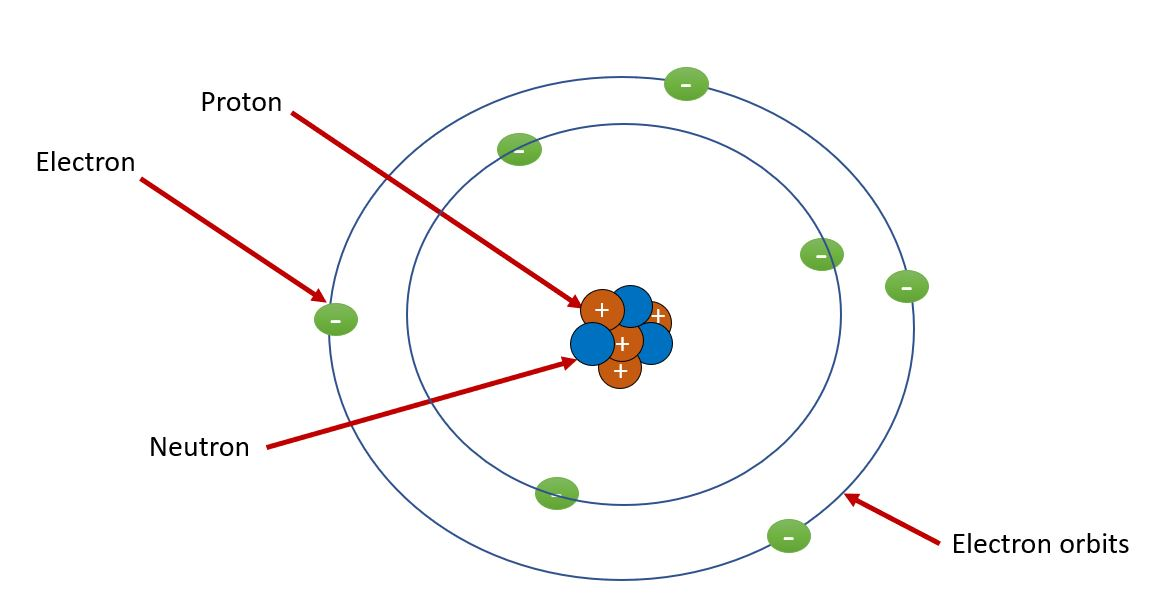
Electron configuration is a cornerstone of modern chemistry. It explains why elements behave the way they do — why they bond, how they react, and even why the periodic table looks the way it does.
In this post, we’ll explore how electrons are arranged in atoms, step-by-step, using four key principles:
-
1.4.1 The Aufbau Principle
-
1.4.2 The Pauli Exclusion Principle
-
1.4.3 Hund’s Rule
-
1.4.4 Condensed Electron Configurations
Each section includes examples, explanations, and practice questions to help you master the topic for your exam.
What Is Electron Configuration?
Electron configuration is the arrangement of electrons in atomic orbitals (s, p, d, f) according to specific rules of quantum mechanics. These configurations determine:
-
The chemical properties of elements
-
Their positions in the periodic table
-
Their bonding behavior
Atoms have energy levels (shells), each of which contains sublevels (s, p, d, f), which are made up of orbitals. Each orbital holds a maximum of 2 electrons.
Atomic Structure: Energy Levels, Sublevels, and Orbitals Explained
To understand how electrons are arranged in an atom, we need to look at the three key layers of atomic structure:
-
Energy Levels (Shells)
-
Sublevels (Subshells)
-
Orbitals
Let’s break them down step by step.
Energy Levels (Shells)
Atoms have energy levels, also called electron shells, which are designated by the principal quantum number (n = 1, 2, 3, 4…).
-
n = 1 is the first shell (closest to the nucleus), and it has the lowest energy.
-
As n increases, the shell is farther from the nucleus and has higher energy.
-
Each energy level can hold a maximum number of electrons, given by the formula:
Maximum electrons per shell = 2n²

Sublevels (Subshells)
Each energy level is made up of sublevels, which define the shape of the region where electrons are likely to be found. There are four main sublevels:



-
Sublevels are designated as 1s, 2s, 2p, 3d, etc.
-
The number of orbitals increases as the sublevel becomes more complex.
Orbitals
An orbital is a region in space where there is a high probability of finding an electron.
-
Each orbital can hold a maximum of 2 electrons, and they must have opposite spins (↑↓) due to the Pauli Exclusion Principle.
-
Orbitals come in sets depending on the sublevel:

💡 Orbitals are not fixed paths like planetary orbits, but rather cloud-like regions based on probability.
Putting It All Together
Let’s take oxygen (Z = 8) as an example:
-
Total electrons: 8
-
Electron configuration: 1s² 2s² 2p⁴
Breakdown:
-
1s²: First shell (n=1), s sublevel → 1 orbital → holds 2 electrons
-
2s²: Second shell, s sublevel → 1 orbital → holds 2 electrons
-
2p⁴: Second shell, p sublevel → 3 orbitals → holds 4 electrons (2 orbitals fully filled, 1 half-filled)
The Four Quantum Numbers – Quick Overview
Each electron in an atom is uniquely identified by a set of four quantum numbers:
1. Principal Quantum Number (n)
-
Indicates the main energy level or shell.
-
Values: 1, 2, 3…
-
Higher n = farther from nucleus + more energy.
2. Angular Momentum Quantum Number (l)
-
Defines the sublevel shape (s, p, d, f).
-
Values: 0 to (n–1)
-
l = 0 → s
-
l = 1 → p
-
l = 2 → d
-
l = 3 → f
-
3. Magnetic Quantum Number (ml)
-
Tells the orientation of the orbital in space.
-
Values: –l to +l
-
Example: for p (l = 1), ml = –1, 0, +1
-
4. Spin Quantum Number (ms)
-
Describes the direction of electron spin.
-
Values: +½ or –½
-
Each orbital holds max 2 electrons with opposite spins.
1.4.1 The Aufbau Principle – “Building Up”
Definition: Electrons fill the lowest available energy levels before occupying higher ones.
This filling order is not simply based on shell number (n), but rather on energy. For example, 4s has lower energy than 3d, so it fills first.
Electron Filling Order:
1s → 2s → 2p → 3s → 3p → 4s → 3d → 4p → 5s → 4d → 5p → 6s → 4f → 5d → 6p…
you can draw the following figure and get this electron filling order easily for the exam.

Example:
Phosphorus (Z = 15)
→ Configuration: 1s² 2s² 2p⁶ 3s² 3p³
Electrons fill the orbitals in the correct energy order.
✅ Practice Questions:
-
Write the electron configuration of aluminum (Z = 13)
➤ Answer: 1s² 2s² 2p⁶ 3s² 3p¹ -
Which orbital is filled first: 4s or 3d?
➤ Answer: 4s is filled before 3d
The Pauli Exclusion Principle – “No Sharing the Same Seat!”
Definition: No two electrons in an atom can have the same set of quantum numbers. So, each orbital holds a maximum of 2 electrons, and they must have opposite spins.
Opposite Spins:
We represent this with ↑ and ↓.
For example, a filled 1s orbital: ↑↓
This rule helps explain why orbitals are limited in capacity and why electron spin is important.
✅ Practice Questions:
-
How many electrons can occupy the 3p sublevel?
➤ Answer: 6 electrons (3 orbitals × 2 electrons each) -
Can two electrons in the same orbital have the same spin?
➤ Answer: No, they must have opposite spins.
Hund’s Rule – “Spread Out Before You Pair Up”
Definition: When filling orbitals of equal energy (e.g., three p orbitals), electrons fill each orbital singly with the same spin before pairing.
This reduces electron repulsion and increases atomic stability.
Example – Nitrogen (Z = 7):

Configuration: 1s² 2s² 2p³
→ 2p orbitals: ↑ ↑ ↑
Each 2p orbital has one electron before any pairs are formed.
✅ Practice Questions:
-
Which rule explains why the three 2p orbitals are each singly occupied before pairing?
➤ Answer: Hund’s Rule -
Draw the orbital diagram for oxygen (Z = 8).
➤ Answer:
-
1s: ↑↓
-
2s: ↑↓
-
2p: ↑↓ ↑ ↑
Condensed Electron Configurations – Noble Gas Notation
Definition: A shorthand notation using the previous noble gas to represent the inner electron configuration.
This method saves time and space, especially for larger atoms.
Examples:
-
Chlorine (Z = 17):
Full: 1s² 2s² 2p⁶ 3s² 3p⁵
Condensed: [Ne] 3s² 3p⁵ -
Iron (Z = 26):
Full: 1s² 2s² 2p⁶ 3s² 3p⁶ 4s² 3d⁶
Condensed: [Ar] 4s² 3d⁶
💡 Use the noble gas from the previous period.
✅ Practice Questions:
-
Write the condensed configuration for calcium (Z = 20)
➤ Answer: [Ar] 4s² -
What is the full and condensed electron configuration of sulfur (Z = 16)?
➤ Full: 1s² 2s² 2p⁶ 3s² 3p⁴
➤ Condensed: [Ne] 3s² 3p⁴
Common Exceptions in Electron Configurations
Some transition metals don’t follow the expected pattern:
-
Chromium (Z = 24)
Expected: [Ar] 4s² 3d⁴
Actual: [Ar] 4s¹ 3d⁵ (more stable half-filled d-subshell) -
Copper (Z = 29)
Expected: [Ar] 4s² 3d⁹
Actual: [Ar] 4s¹ 3d¹⁰ (more stable full d-subshell)
⚠️ These exceptions are important for exams!
Summary Table

🎯 Final Practice Questions
-
Which of the following electron configurations is correct for magnesium (Z = 12)?
A) 1s² 2s² 2p⁶ 3s²
B) 1s² 2s² 2p⁶ 3p²
C) 1s² 2s² 2p⁴ 3s²
➤ Answer: A -
What is the electron configuration of Zn²⁺? (Zinc, Z = 30)
➤ Neutral Zn: [Ar] 4s² 3d¹⁰
➤ Zn²⁺: [Ar] 3d¹⁰ (loses 2 electrons from 4s) -
Explain why 4s fills before 3d, but 3d is written after 4s in the configuration.
➤ Answer: 4s has lower energy when filling, but once filled, 3d becomes lower in energy and is written after 4s.