In the chemical processing industry, safety is not just a regulatory requirement—it’s a lifeline. Chemical plants deal with hazardous substances, high pressures, extreme temperatures, and reactive systems that can pose serious threats if not managed properly. A single lapse can lead to catastrophic consequences, affecting people, the environment, and business sustainability.
This guide offers a comprehensive learning structure and explains the critical safety systems, hazards, tools, and best practices every chemical engineer, technician, or plant operator should know.
Learning Structure for Plant Safety
To build a strong safety culture, you need a structured educational approach. Below is a framework that guides safety training and competency development in chemical industries.
1. Introduction to Plant Safety
-
Why Safety Matters:
Safety is the foundation of sustainable operations. It prevents accidents, protects employees, preserves assets, and enhances plant efficiency. -
Basic Safety Terminology:
Terms like hazard, risk, near-miss, incident, exposure, and mitigation must be well understood to foster clear communication.
Hazard – A potential source of harm or danger.
Example: A flammable liquid stored near a heat source.
Risk – The chance that a hazard will cause harm, considering the likelihood and severity.
Example: The risk of fire increases if the flammable liquid is not stored properly.
Near-Miss – An unplanned event that did not cause harm but had the potential to do so.
Example: A worker slips but regains balance and avoids a fall.
Incident – An event that causes or could cause harm, injury, damage, or loss.
Example: A chemical spill that causes skin burns or equipment damage.
Exposure – The contact or presence of a person with a hazard (chemical, physical, or biological).
Example: Inhaling solvent vapors or touching a hot surface.
Mitigation – Actions taken to reduce the severity or impact of a hazard or incident.
Example: Installing gas detectors and emergency shutdown systems.
-
Types of Hazards:

2. Hazard Identification and Risk Assessment
-
Methods of Hazard Identification:
Use checklists, job safety analysis (JSA), audits, and inspections to detect hazards before they cause harm. -
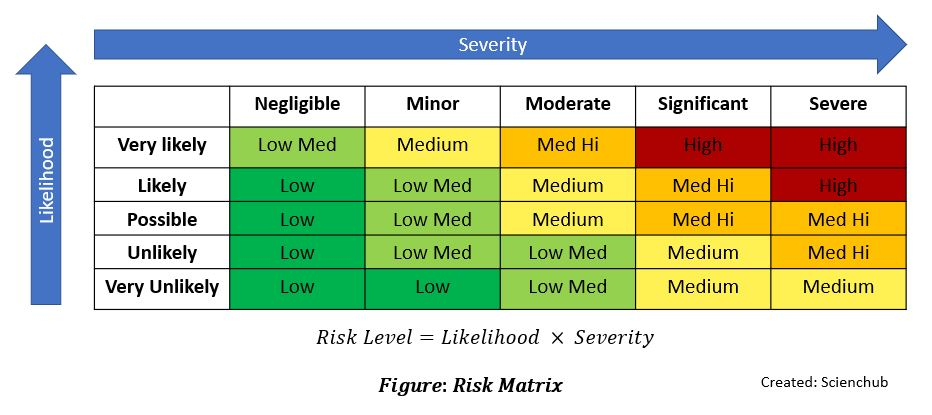
Risk Assessment Process:
Evaluate likelihood × severity to prioritize controls.


-
Hierarchy of Controls:
1. Elimination
Remove the hazard entirely from the workplace.
Example: Replace a toxic chemical process with a safer method.
2. Substitution
Replace the hazard with a less dangerous one.
Example: Use a less volatile solvent.
3. Engineering Controls
Physically isolate people from the hazard.
Example: Enclose equipment, install ventilation or interlocks.
4. Administrative Controls
Change how people work to reduce exposure.
Example: Training, shift rotations, signage, SOPs.
5. Personal Protective Equipment (PPE)
Last line of defense—protect the worker.
Example: Gloves, goggles, respirators.

3. Safety Policies, Procedures, and Standards
-
Developing Policies:
Establish clear goals, responsibilities, and enforcement mechanisms. -
Roles and Responsibilities:
-
Management: Resource allocation and accountability
-
Supervisors: Daily oversight and team training
-
Employees: Safe conduct, reporting, and cooperation
-
-
Safety Management Systems (SMS):
Frameworks like ISO 45001 integrate safety into daily operations. -
Legal Compliance:
Adhere to OSHA, EPA, and local regulations to avoid fines and improve risk control.
4. Personal Protective Equipment (PPE)
PPE is vital for individual protection when other controls cannot eliminate risks.
-
Types of PPE:

-
Selection and Use:
Based on hazard type and job requirement. Must be inspected regularly and fitted correctly. -
Limitations:
PPE does not eliminate hazards, only shields workers.
5. Machine and Equipment Safety
-
Guarding & Interlocks:
Protect operators from moving parts and accidental contact.
-
Lockout/Tagout (LOTO):
Ensures complete isolation of energy sources during maintenance.

-
Standard Operating Procedures (SOPs):
Documented safe practices for routine and emergency operations. -
Inspection & Maintenance:
Preventive upkeep of machinery avoids mechanical failures and unexpected downtime.
6. Chemical Safety
-
Material Safety Data Sheets (MSDS / SDS):
Provide information on handling, exposure, first aid, storage, and reactivity. -
Safe Handling and Storage:
Segregate incompatible chemicals, use proper containers, and label everything clearly. -
Spill Control:
Include containment trays, spill kits, and trained response teams. -
Hazard Communication:
Use signage, labels, and visual cues throughout the plant.
7. Emergency Preparedness and Response
-
Types of Emergencies:
Fire, explosion, chemical leaks, toxic gas release, medical emergencies -
Emergency Action Plans (EAP):
Step-by-step plans for evacuation, containment, and communication -
Evacuation Routes & Muster Points:
Clearly marked and regularly drilled -
Firefighting Systems:
Extinguishers, fire blankets, deluge systems, hydrants -
First Aid:
CPR-trained staff, stocked first-aid kits, and emergency eyewash stations
8. Safety Training and Competency
-
Needs Assessment:
Identify gaps based on job roles and site risks. -
Training Delivery:
Blended learning with classroom, online, and on-the-job methods -
Safety Culture:
Promote accountability, communication, and proactive behavior. -
Competency Testing:
Verify understanding through assessments, certifications, or simulations.
9. Environmental Safety and Health
-
Waste Management:
Segregation, treatment, and disposal of hazardous and non-hazardous waste -
Pollution Prevention:
Emission control, water conservation, solvent recovery -
Health Hazards:
Address noise, vibration, temperature, confined space entry -
Ergonomics:
Design tasks to minimize repetitive stress and fatigue
10. Incident Reporting and Investigation
-
Reporting Procedures:
Encourage prompt reporting of incidents and near misses -
Root Cause Analysis (RCA):
Use 5 Whys, Fishbone Diagrams to uncover systemic causes


-
Corrective/Preventive Actions (CAPA):
Implement changes and monitor results -
Performance Metrics:
Track KPIs like TRIR, LTIR, near miss frequency
11. Continuous Improvement in Safety
-
Audits & Inspections:
Conduct regular reviews of systems and equipment -
Safety Committees:
Engage employees in identifying problems and solutions -
Feedback Loops:
Listen to workers—they often spot issues early -
Use of Technology:
Drones for inspection, SCADA systems, AI monitoring
Why Safety Is Essential in Chemical Plants
Without robust safety protocols, chemical plants can face:
-
Fires and Explosions
-
Toxic Releases
-
Operational Downtime
-
Injuries or Fatalities
-
Environmental Damage
-
Reputational and Legal Consequences
These risks make safety not just ethical—but essential for business continuity.
Common Hazards in the Chemical Industry
-
Flammable gases and liquids
-
Reactive chemicals
-
High pressure vessels
-
Toxic substances
-
Mechanical hazards from rotating or pressurized equipment
-
Ergonomic stress and fatigue
-
Electrical hazards and arc flashes
Safety Tools and Systems in Chemical Plants
Chemical plants are protected by multiple layers of safety systems, categorized as:
1. Process Safety Systems
-
Pressure Relief Devices (PRDs): Prevent vessel ruptures
-
Emergency Shutdown Systems (ESDs): Automated equipment shutdown
-
Safety Instrumented Systems (SIS): Perform critical actions like closing valves or tripping pumps
-
Inert Gas Blanketing: Suppresses flammable vapor formation
-
Explosion Vents/Flame Arrestors: Release pressure and contain ignition
2. Personal Protective Equipment (PPE)
-
Chemical suits, gloves, respirators, eye and face protection
3. Gas and Fire Detection Systems
-
Fixed Detectors: For H₂S, CH₄, VOCs
-
Portable Monitors: For confined space entry
-
Smoke/Flame Detectors: Trigger suppression systems
4. Fire and Explosion Protection Systems

5. Control and Monitoring Systems
-
DCS (Distributed Control System): Manages process plant
-
PLC (Programmable Logic Controller): Controls specific units
-
SCADA: Supervises remote systems
-
Alarms: Alert deviations in pressure, temperature, flow, etc.
6. Emergency Response and Escape Systems

Best Practices for Safety in Chemical Plants
-
Conduct Process Hazard Analyses (PHA): HAZOP, What-If, Fault Tree
-
Follow Permit-to-Work Systems: Hot work, confined space, electrical
-
Standard Operating Procedures (SOPs): Well-documented and reviewed
-
Train Employees Regularly: Safety drills, PPE use, hazard awareness
-
Preventive Maintenance: Equipment, valves, sensors
-
Plan and Practice Emergencies: Evacuation, communication, firefighting
Regulatory Compliance
Plants must comply with:

Compliance ensures legal operation and promotes industry best practices.
Final Thoughts
Safety in chemical plants is not optional—it is mission-critical. An effective safety strategy protects lives, prevents environmental disasters, enhances efficiency, and ensures long-term profitability.
With well-structured learning, smart systems, and a proactive culture, zero incidents is not a dream—it’s an achievable standard.
“You can’t manufacture safety—but you can engineer it into every step.”