Atoms are the basic building blocks of matter. But our understanding of the atom has evolved dramatically over time. Scientists didn’t always know what atoms looked like — they had to experiment, observe, and revise their ideas based on new evidence. Let’s explore the key atomic models that shaped modern chemistry and are essential for A/L Chemistry students.
1. Dalton’s Atomic Theory (1803)

Model Name: Golf Ball Model
John Dalton proposed the first scientific model of the atom:
-
Atoms are tiny, indivisible particles.
-
All atoms of the same element are identical.
-
Atoms of different elements vary in mass and properties.
-
Atoms rearrange in chemical reactions, but they are not created or destroyed.
Limitation: It could not explain internal structures like electrons, or isotopes (atoms of the same element with different masses).
2. Thomson’s Plum Pudding Model (1897)

After discovering the electron through cathode ray experiments, Thomson proposed that:
-
The atom is a uniformly positive sphere (the “pudding”) with negatively charged electrons (the “plums”) scattered throughout.
-
The atom as a whole is neutral, as the negative and positive charges cancel out.
🔹 Limitation: Could not explain the results of Rutherford’s gold foil experiment, which showed that most of the atom is empty space, and the positive charge is concentrated in a small region (the nucleus).
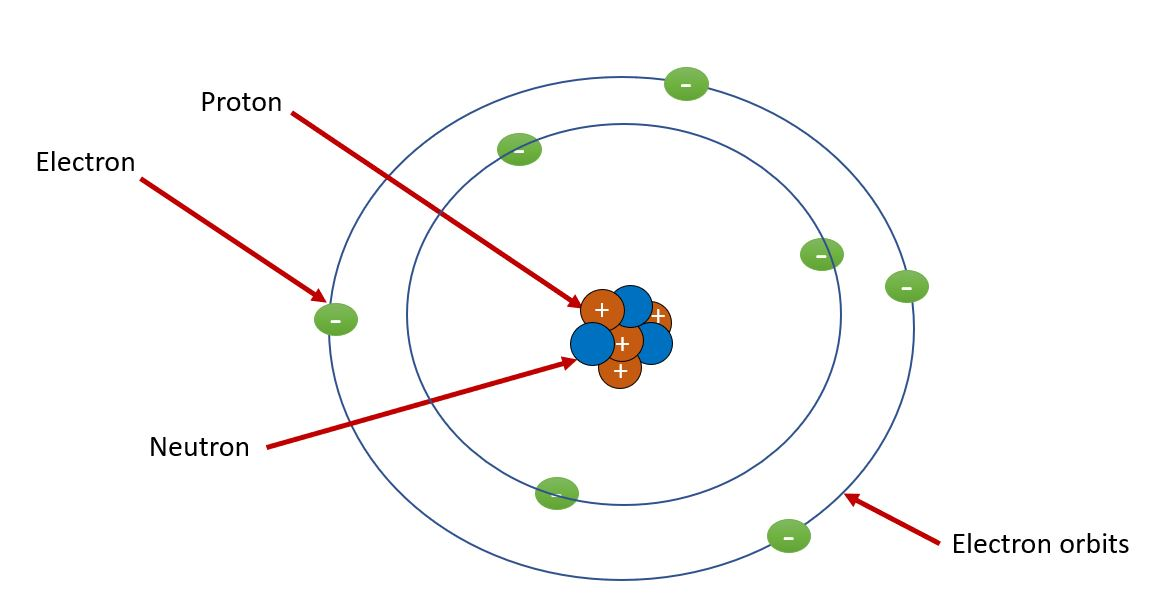
3. Rutherford’s Nuclear Model (1911)

Through his gold foil experiment, Rutherford observed that most alpha particles passed straight through the foil, but some were deflected at large angles. He concluded:
-
The atom is mostly empty space.
-
It has a small, dense, positively charged nucleus at the center.
-
Electrons orbit this nucleus, similar to how planets orbit the sun.
🔹 Limitation: According to classical physics, orbiting electrons should lose energy and spiral into the nucleus. Yet, atoms are stable — something this model couldn’t explain.
4. Bohr’s Model (1913)

Niels Bohr built on Rutherford’s model using ideas from quantum theory. He proposed:
-
Electrons orbit the nucleus in discrete energy levels (shells) without radiating energy.
-
Electrons can jump between levels by absorbing or emitting a fixed amount of energy (a quantum).
-
This model successfully explained the line emission spectrum of hydrogen.
🔹 Limitation: It worked well only for hydrogen and single-electron systems. Could not accurately predict spectra for multi-electron atoms.
5. Quantum Mechanical Model (Modern Model)
The current model of the atom is based on quantum mechanics. It describes atoms more accurately by treating electrons as both particles and waves.
Key features:
-
Electrons are found in orbitals, not fixed paths. These are regions where electrons are likely to be found.
-
Schrödinger’s Equation is used to describe these orbitals, but for A/L, only a conceptual understanding is needed — no math required.
-
The Heisenberg Uncertainty Principle says we cannot know both the exact position and momentum of an electron at the same time — this highlights the probabilistic nature of electron behavior.
-
Wave-particle duality means electrons behave both like particles and waves, a concept crucial to understanding modern atomic structure.
This model provides a much more accurate and detailed view of atoms, especially for multi-electron elements.
Summary Table of Atomic Models